Laboratory of Lymphoma Tumor Biology
Our laboratory was established in January 2018
Primary focus
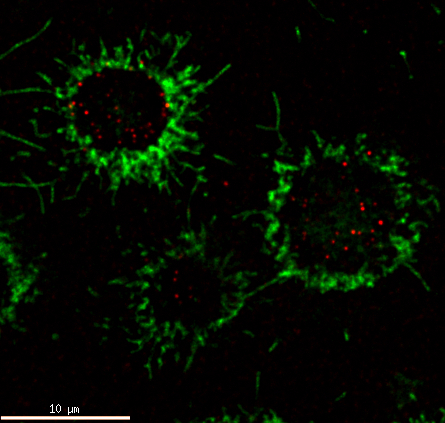
Non-Hodgkin lymphomas are a heterogenous group of hematologic malignancies derived in more than 90% from B-lymphocytes, one subtype of white blood cells. B-cells are part of adaptive immune system and one of their main functions is to recognize antigens and differentiate into plasmatic cells to produce antibodies. Hallmark molecule of B-cells is B-cell receptor (BCR), which is specifically responsible for antigen binding and recognition. Majority of B-cell derived malignancies retain surface expression of BCR, which is actively signaling and supports growth and survival of tumor cells. Similarly, as is heterogonous group of lymphomas, differences exist also in types of signaling it provides. In some lymphomas, BCR signals through “chronic active” type of BCR signaling that resembles normal signaling after antigen binding, in other lymphomas, BCR signaling resembles “tonic” type of signaling that normally provides basic signal necessary for survival of mature B-cells. However, many details and differences in BCR signal initiation and its transmission to individual signal mediators are not fully characterized. Inhibition of BCR signaling and its related signal mediators is also one of novel strategies of lymphoma treatment, but more information is needed to identify novel therapeutic targets or personalize use of existing inhibitors.
Latest developments in technology and related research resulted in unprecedented advancement in our understanding of mechanisms leading to lymphomagenesis, however, many mechanisms of aberrant signaling, metabolic changes, transcription regulation, or interactions of tumor cells with tumor microenvironment are still not clear.
Using advanced methods for targeted genomic modifications, protein-protein interactions detection, high-resolution microscopy, omics methods (proteomics, genomics, and metabolomics), in vivo models, or intracellular biosensors, the general aim of our group is to address missing information about cellular processes critical for lymphoma development, tumor cell growth, and contribute to our basic understanding of these processes in general.
Specific Projects:
Molecular details of B-cell receptor signal initiation and its transmission to protein kinase B.
Identification of biomarkers for personalized use of B-cell receptor signaling inhibitors.
Description of the role of lymphoma specific genomic alterations of novel epigenetic regulators.
Mechanisms of oncogenic PI3K/AKT pathway activation.
Tumor specific crosstalk between signaling, metabolism and cell cycle.
Specifics of tumor metabolism.
Circulating tumor DNA as a diagnostic and therapy prediction tool.
Our group profile at the BIOCEV website